Thoracic trauma
Chest wall stabilization
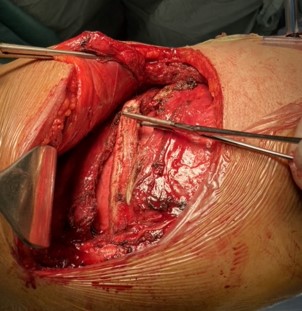
Rib fractures are by far the most common consequence of chest trauma, even when minor. The presence of multiple rib fractures, sometimes fragmented and/or displaced, sometimes with fragments protruding into the pleural cavity, or the presence of flail chest, cause intense chest pain and significantly affect the respiratory dynamics of the patient. In particular, they can cause severe respiratory failure, often requiring intubation and prolonged hospitalisation in intensive care unit. In addition, impaired respiratory dynamics lead to shallow breathing and of bronchial secretions retention, with a very high risk of pneumonia development.
In recent years, chest wall stabilisation surgery has developed, especially in the United States. The advantages of this surgery are: a) significant reduction in chest pain, b) improvement in respiratory dynamics, c) the possibility of extubating the patient more quickly, allowing for earlier discharge from intensive care, and d) much faster functional recovery, with a quicker return to daily life and work.
In the last three years, the Thoracic Surgery Unit in Baggiovara has gained significant experience in this surgery, having performed more than 80 complex procedures, making it the most experienced unit in Italy in this field and one of the most advanced in Europe.
Clinical research is now focused on two main areas: the assessment of clinical factors predictive of possible post-operative complications and the development of a protocol for the multidisciplinary management of trauma patients, involving all professionals directly involved in the immediate management of trauma.
Head of research: Prof. Alessandro Stefani
Thoracic oncology
Surgical treatment of Locally Advanced Lung tumors after induction chemo-immunotherapy
Locally advanced primary lung cancers (Stages IIb-IIIa) account for approximately 30% of new diagnoses per year. The correct indication for their treatment has been the subject of controversy among oncologists, radiotherapists and thoracic surgeons for years. Recent developments in oncology, with the introduction of clinical trials involving immunotherapy drugs, have undoubtedly increased the treatment options for these neoplasms, increasingly involving surgeons in order to improve clinical response and patient survival.
In multidisciplinary clinical discussions for these tumours, important new insights are coming up: a) the importance of proper clinical/invasive staging of the tumour; b) rethinking the idea of surgical resectability of the lesion (a judgement that should only be made by the thoracic surgeon); c) the assessment of resectability (the ability to achieve radical oncological resection) and the patient’s operability (the ability to tolerate surgery and overcome any post-operative complications).Surgery after chemo-immunotherapy is very complex and requires significant expertise.
The cause of this technical complexity is linked to the profound tissue alterations induced by cancer treatment (and in particular by immunotherapy). There are few centres in Italy and Europe capable of performing this type of surgery. Over the last year, thanks to close collaboration with oncologists, the Thoracic Surgery Unit has performed 14 surgical procedures on patients treated with induction chemo-immunotherapy. This series, achieved in such a short time, places it among the most important Italian units.
The aims of clinical research in this field are: a) definition of a protocol for identifying potential patients for combined treatment; b) identification of clinical predictors capable of predicting a clinical/pathological response to combined therapy; c) defining a clinical protocol for the prevention and possible management of post-operative complications; d) developing a clinical follow-up protocol for these patients; e) defining an Italian database of locally advanced neoplasms treated with induction chemo-immunotherapy.
Head of research: Prof. Alessandro Stefani
Salvage Surgery after chemo-immuno-radiotherapy for Locally Advanced Lung tumours (Salvage Surgery)
The treatment of patients with locally advanced lung cancer is still widely debated, especially in light of recent therapeutic innovations resulting from the development of immunotherapy. For tumours deemed unresectable upfront, the treatment options are chemotherapy (± immunotherapy) and radiotherapy. The recent introduction of clinical protocols for radiotherapy combined with immunotherapy (PACIFIC trial) has significantly increased the 3-year survival rate of these patients. However, the problem of disease recurrence or metastases still affects more than 30% of patients.
Salvage surgery is considered in well-defined clinical situations: a) recurrence of disease after an initial response to the treatment; b) persistence of disease after a clinical/radiological restaging has determined that the lesion is resectable; c) presence of treatment-related complications (bronchial fistulas, lung abscesses, haemoptysis). The ultimate goal of this surgery is to increase local control of the disease, thereby also increasing patient survival.
From a surgical point of view, these are extremely complex surgical procedures due to the direct effects of radiotherapy and immunotherapy, which effectively disrupt the hilar structures, causing profound tissue alterations. At the same time, the surgical procedures are anatomically very extensive and complex, involving large amounts of tissue resection, with extended pneumonectomy being the most common. The post-operative course of these patients is also often complex, due to the possible development of complications, among which respiratory ones are the most serious and threatening.
Over the last three years, the Thoracic Surgery UNIT in Baggiovara has performed several salvage surgeries for advanced lung cancer, treated with exclusive chemo-radio-immunotherapy, making it one of the most experienced Italian units in this field.
Clinical research projects are focused on increasing multidisciplinary collaboration between oncologists, radiotherapists, thoracic surgeons, and pulmonologists to reach a consensus on clinical indications for life-saving surgery, implementing clinical collaboration with anaesthetists, intensivists, pulmonologists, cardiologists, and physiatrists in the post-operative management of these complex and fragile patients, with the aim of significantly reducing possible complications, and finally, developing a clinical database to collect the experiences of other centres.
Head of research: Prof. Pier Luigi Filosso
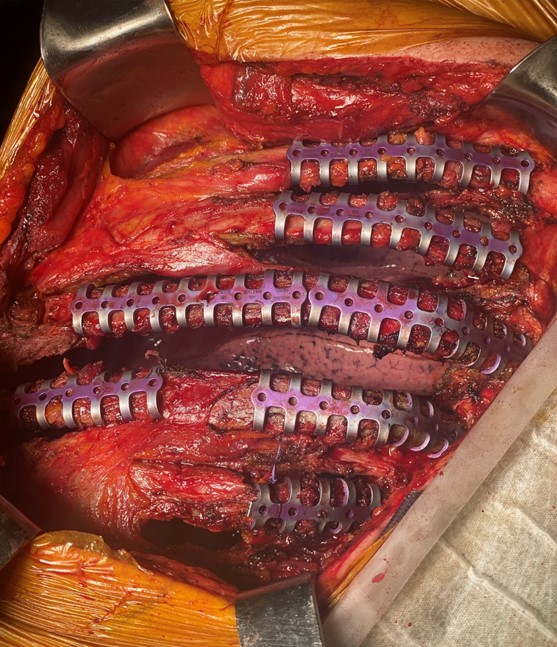
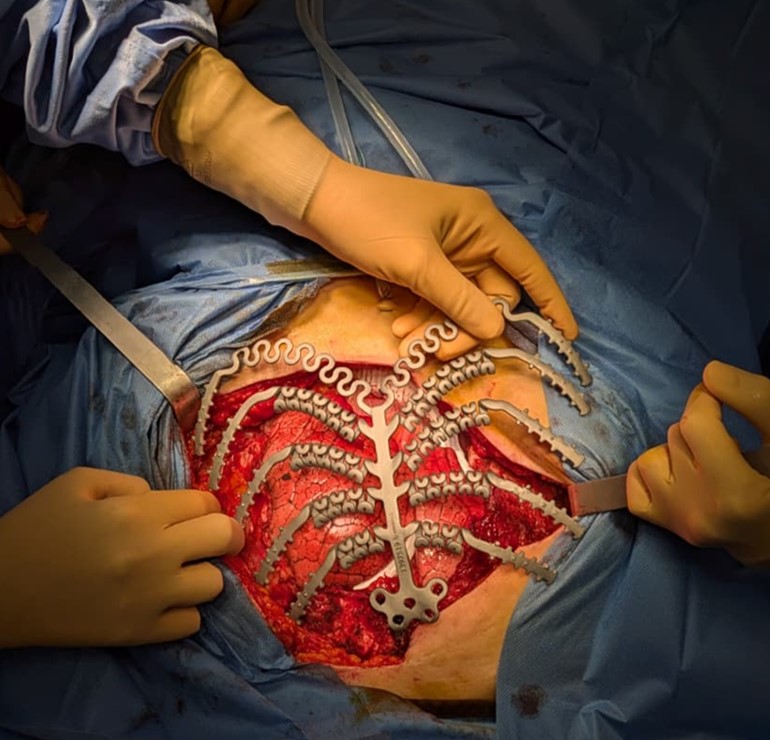
Surgical Treatment of Chest Wall Tumours
Malignant tumours of the chest wall are extremely rare and may arise from primary bone tumours or bone metastases from other tumours (e.g. breast, ovary, uterus, lung). The complexity of their treatment depends on: a) their rarity (which makes it difficult to establish a general treatment protocol); b) their possible extensiveness (which generally requires very wide surgical resection); c) the difficulty of reconstruction after large resection and d) possible post-operative complications.Particular attention must be given during the reconstruction to restoring wall rigidity, using titanium prostheses that may even be created “ad hoc” for the patient, based on pre-operative radiographic studies (CT or MRI).
Active collaboration with colleagues from the Plastic Surgery Unit in Modena Policlinico makes it possible to perform the reconstructive phase in the best possible manner, with adequate muscle transposition to cover the prosthesis, with the aim of avoiding its possible infections.Over the last three years, the Thoracic Surgery Unit in Baggiovara has performed several interventions for tumours of the chest wall. Of particular interest were two recent sternum replacements with titanium prostheses, for mediastinal seminoma and ovarian tumour metastases.The clinical research project for these pathologies involves increasingly close collaboration with oncologists and radiotherapists in order to standardise the clinical treatment protocol. Similarly important is the collaboration with companies that produce titanium prostheses, with the aim of studying the clinical effects of prosthesis placement on the chest wall over time.
Research supervisor: Prof. Pier Luigi Filosso
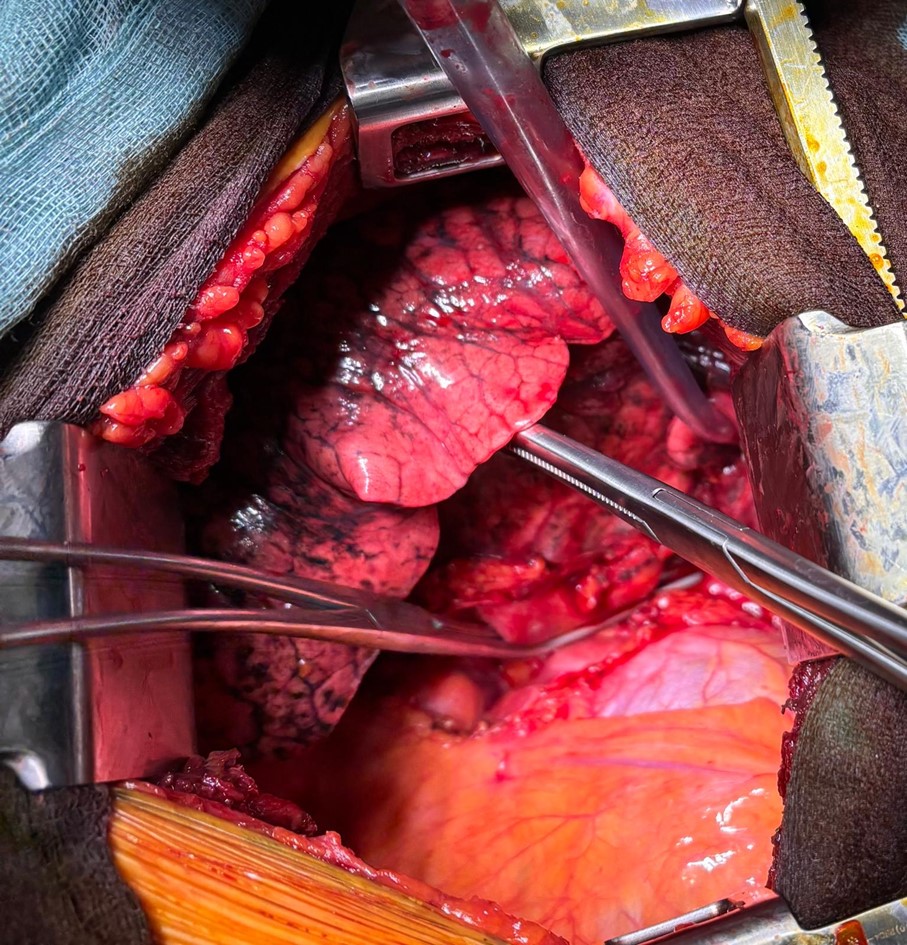
Locally advanced thymomas and thymic carcinomas
Thymomas (T) and thymic carcinomas (CT) are very rare primary neoplasms of the anterior mediastinum, with an incidence of 0.13 cases per 100,000 people/year. In most cases (and especially for T), diagnosis occurs at an early stage. They can sometimes be associated with paraneoplastic syndromes, of which myasthenia gravis (MG) is the most common.
However, in 7-11% of cases, the tumour is diagnosed in a more invasive form (especially in TC), with possible invasion of the pleura, pericardium, lung, or directly the adjacent anatomical structures (especially the great vessels). Even after radical surgery (R0), recurrence is possible in 30-35% of cases. The optimal treatment of locally advanced thymic neoplasms requires a multidisciplinary approach involving the oncologist, radiotherapist, thoracic surgeon, pulmonologist, and pathologist.
Surgery, especially radical , remains the treatment of choice even for the most aggressive and advanced tumours. In addition, patients with these types of cancer are generally younger and in better condition than those with lung or oesophageal cancer, and are therefore more able to tolerate combined chemo-radiotherapy treatments and/or more complex procedures.Patients with advanced neoplasms are generally treated with induction chemotherapy (usually 4 cycles ), followed by surgery and, if necessary, radiotherapy (in the event of invasion of adjacent structures or residual disease).
In case of locoregional recurrence, upfront surgery is often indicated, especially if it is deemed feasible with oncological radicality.In cases of locally advanced lesions and/or recurrence after previous surgery, surgery is complex, requiring the possible resection of various thoracic structures and sometimes a combined approach with other specialist surgical teams. It also requires significant involvement of anaesthetists, who are involved in the difficult pre-, intra- and post-operative management of these patients.
In the last three years, the Thoracic Surgery Unit in Baggiovara has treated a large number of patients with advanced/recurrent T or TC, sometimes in collaboration with cardiac surgeons from the Hesperia Hospital in Modena, making it one of the Italian thoracic surgery units with the most clinical experience in this field. The current clinical research project plans to further increase the number of such patients and, at the same time, to deepen collaboration with oncologists and radiotherapists in order to standardize the treatment protocol for these diseases and increase the long-term survival of these patients.
Research supervisor: Prof. Pier Luigi Filosso
Generation of a 3d model of the lung for the treatment of pulmonary fibrosis
The Pulmonary Fibrosis diseases are characterized by a pathological activation of fibroblasts and by an excessive deposition of extracellular matrix components in the lung. For now, only two drugs have been approved, Nintedanib and Pirfenidone, which could slow down the fibrotic progression but are not able to arrest this condition. The lack of therapeutic strategies is partly due to poor predicting power of the preclinical models. To date, Precision Cut Lung Slices (PCLS) represent the most predictive model for testing new antifibrotic drugs. In parallel, 3D bioprinting is emerging as a promising technology to mimic fibrotic disease in a controlled and reproducible manner.
The aim of this study is to develop advanced in vitro models that closely mimic pathological conditions by integrating biomimetic tissue constructs with pharmacological modulation. These models are designed to enable disease simulation and serve as robust platforms for drug screening.
For this study, the hPCLS (human Precision-Cut Lung Slices) approach was employed using biopsies obtained from healthy donors. In parallel, a 3D bioprinting approach was utilized, employing cells derived from fibrotic human lung tissue to create disease-mimicking models. These platforms constructed were then subjected to various drug treatments to evaluate therapeutic responses.
In this study, we propose two different and advanced techniques able to replicate the clinical features of fibrosis with remarkable similarity and complexity. Both models were treated by two different stimuli : TGFβ and PFC which induced two different levels of clinical status. Once induced the fibrotic pathology, we tested two different drugs: the commercial Nintedanib (NTD) and a new drug candidate (cmpB) to evaluate the ability of the models to revert the clinical features of disease. In details, we studied the microarchitecture of hPCLS and Bioprinted models by SEM and imaging analysis to highlight both ECM deposition, matrix remodelling, alveolar structure and cell behaviour. All outcomes are supported by quantifiable parameters suitable for numerical and statistical assessment.
Human PCLS (Precision-Cut Lung Slices) retain the native lung structure and cell diversity, offering a highly predictive ex vivo model. • 3D bioprinting allows the creation of complex, human- like tissues using living cells , a game changer for drug testing and disease modelling. • Both models are able to red uce the pathological features of fibrosis after drug treatments, supporting the use of the approaches as platform for testing. • The development of new models represents a crucial step toward a future focused on advanced screening platforms that more accurately reflect the complexity of human diseases, enabling more effective responses to emerging therapeutic challenges.
Research supervisors: Prof. Pier Luigi Filosso& Dott.ssa Tiziana Petrachi
